Histopathology Lab Workflow Redesign
Lab technicians, pathologists, and micro dissection specialists relied on spreadsheets, manual queues, and fragmented systems to process tissue samples. I led shadowing sessions and UAT to design a unified system that replaced manual work across the entire histopathology pipeline.
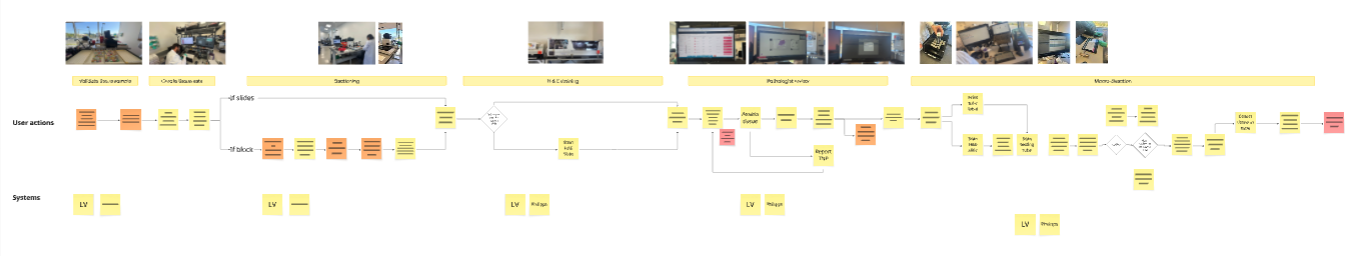
The lab problem: Natera’s histopathology workflow — from tissue validation through micro dissection — spanned five major stages, each with its own tools, manual handoffs, and pain points. Lab technicians toggled between LabVantage (LV) for accessioning, Philips for digital pathology, and spreadsheets for everything in between.
Why this matters: Every manual step in histopathology introduces risk. Mislabeled tubes, missed scanning windows, and broken integrations between systems don’t just slow the lab down — they can compromise sample integrity and downstream genetic test results.
The constraint: Design for expert lab users who work with physical specimens, scanners, and microscopes — not just screens. The system had to fit into the physical choreography of the lab, not disrupt it.
I led multiple rounds of contextual inquiry and shadowing sessions with lab technicians, pathologists, and micro dissection specialists in their actual work environments. The goal was to map every action, decision point, and system touchpoint across the full histopathology pipeline.
Methods
- In-lab shadowing sessions observing tissue validation, sectioning, staining, pathologist review, and micro dissection
- Workflow mapping of user actions, system interactions, and decision branches (slides vs. blocks)
- Pain point identification through observation and contextual interviews
- Systems audit of LabVantage (LV) and Philips integration points
The workflow map captured the full end-to-end process: Validate tissue sample → Create tissue sets → Sectioning → H&E staining → Pathologist review → Micro dissection, with branching logic for slides versus blocks at the sectioning stage.
Finding 1: Manual queue management
Lab staff manually added “children” samples to queues — a repetitive task that could be automated through barcode scanning. Every manual entry was a potential error.
Finding 2: Broken system integrations
The integration between Bioanalyzer and LabVantage was incomplete. System-generated codes weren’t automatically added to cases, forcing manual data reconciliation.
Finding 3: Disconnected pathology reports
Path report PDFs weren’t linked to case details in the system. Staff had to retrieve reports from LIMS separately, losing context and time during pathologist review.
Finding 4: Tube-label mismatch risk
Manual matching between cycles of data (tubes vs. labels) during micro dissection created a high-risk error point. Scanning before and after processing had no valid time window for “undo” operations.
Finding 5: Historical scanning gaps
Historical scanning of all tissue samples lacked consistent records, making it difficult to trace case history. Scanning samples initiated lab workflows and retrieved case info, but this process wasn’t standardized.
Design for the physical choreography of the lab. Technicians move between stations, scanners, and microscopes — the system has to meet them where they work, not pull them to a screen.
Design principle
What we built: A unified system replacing spreadsheets and manual tracking across all five histopathology stages. The new workflow automated queue management through barcode scanning, linked pathology reports directly to case records, and consolidated LabVantage and Philips touchpoints into a coherent experience.

Key design decisions
- Scan-driven workflow — barcode scanning triggers automatic queue placement and case retrieval, eliminating manual entry
- Integrated pathology reports — path report PDFs linked directly to case details, leveraging document management
- Automated code generation — system-generated codes added to cases automatically, closing the Bioanalyzer-LV gap
- Undo-safe scanning — defined valid time windows for scan corrections during micro dissection
- Branch-aware UI — interface adapts based on whether the sample follows the slides or blocks path after sectioning
Testing approach: Conducted user acceptance testing (UAT) with lab technicians, pathologists, and micro dissection specialists using the actual system in their lab environments. Tested each workflow stage with real tissue processing scenarios.
What we validated: The scan-driven workflow reduced manual entry points. Integrated pathology reports eliminated the context-switching overhead of retrieving reports from LIMS. The branch-aware interface correctly guided users through slides vs. blocks paths without confusion.
Quantitative impact
- Replaced spreadsheets and manual tracking across all 5 histopathology stages
- Consolidated LabVantage and Philips interactions into a unified workflow
- Eliminated manual queue management through scan-driven automation
- Linked pathology reports directly to case records, removing retrieval overhead
Qualitative impact
- Reduced error risk at tube-label matching and sample handoff points
- Standardized scanning and tracking across the full tissue processing pipeline
- Lab staff no longer context-switch between disconnected systems for a single case
What I learned: Lab workflows are deeply physical. You can’t redesign a histopathology pipeline from a conference room — you have to stand next to the microtome, watch the staining process, and see where the technician reaches for a spreadsheet because the system doesn’t support what they need. The shadowing sessions were the most valuable research investment in this project.
What I’d do differently: I would have mapped the full systems architecture (LV, Philips, Bioanalyzer, LIMS) earlier in discovery. Some integration constraints surfaced late and required design rework that could have been anticipated.